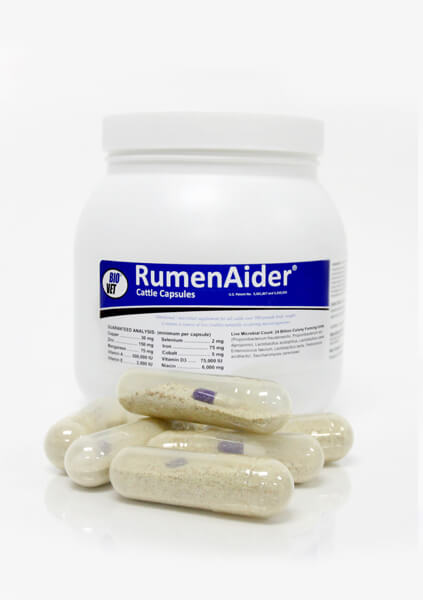
As a leading producer of animal health & nutrition products, Bio-Vet combines the latest research with real-world experience to deliver innovative solutions for your farm. Our unique approach to animal care and production focuses on supporting your animals’ natural system to improve animal health and grow profits.
What Bio-Vet Does
The journey between the health line and death line of your livestock.
What Our Customers Say
Maximize Animal Health
Bio-Vet offers a complete line of products, including capsules, boluses, pastes, soluble powders, calf and cattle electrolytes, and daily feed additives. Our products are key components of a complete nutritional program for livestock. With the help of our experienced veterinarians and production animal professionals, we continue to develop products based on research and industry demand.
Best-Sellers
Our promise to you is to formulate products with the highest quality ingredients to help producers raise healthy animals and grow their profits.